The assessment of food contact materials (FCM) requires knowledge not only of intentionally added substances (IAS), but also of unknown substances that may be hidden in the material.
NIAS in legislation
The so-called NIAS (non-intentionally added substances) are substances that are not intentionally used during the manufacturing process but may inevitably be present in the (finished) material and must therefore also be systematically evaluated to ensure general food safety.
Regulation (EU) No 10/2011 (Plastics Regulation) defines unintentionally introduced substances as ‘an impurity in the substances used, or an intermediate reaction product’ formed during the manufacturing process, or a degradation or reaction product’. At the same time, the Regulation points out that unintentionally introduced substances may be present in materials or articles made of plastics and that the declaration of conformity for the material in question must provide ‘sufficient information on the presence of unintentionally introduced substances’.
Accordingly, it is already assumed here that NIAS may be present in materials, but the assessment of this is the responsibility of the manufacturers/suppliers.
Where do NIAS come from?
The presence of NIAS can have a wide variety of causes and occur along the entire value chain. They are often degradation products of the polymers used (e.g. shorter-chain oligomers, acetaldehyde from PET), oxidation products from additives used (e.g. antioxidants and stabilizers), contamination from the manufacturing process (e.g. lubricants) or the raw materials used, but also recycling-related contributions when using/manufacturing recycled material are possible.
Identification of NIAS
In all cases, the basis for a (risk) assessment is the identification and quantification of the relevant substances. If it is already known in advance or there is a suspicion as to which substances typically occur (or may occur) as NIAS, targeted analysis is possible (e.g. specific migration of acetaldehyde from PET).
However, since NIAS are usually unknown, often unlisted substances with very different chemical properties, the analysis is generally carried out using non-target analysis, i.e. a non-targeted examination (“screening”) of a migration solution of the respective food contact material. Chromatographic separation methods (e.g. gas chromatography, liquid chromatography) are used to separate the complex mixture of substances and quantify it using internal standards.
The substances detected are identified using mass spectrometry, and the data obtained (mass fragmentation) is compared and evaluated using commercial and in-house databases. If a substance cannot be identified more precisely, e.g. because there is insufficient data, its origin must be investigated more closely. It is often helpful to contact the suppliers or take a closer look at the process. Unknown substances are often impurities or by-products or degradation products of the raw materials used, and contamination during the manufacturing process is also a possible source of contamination.
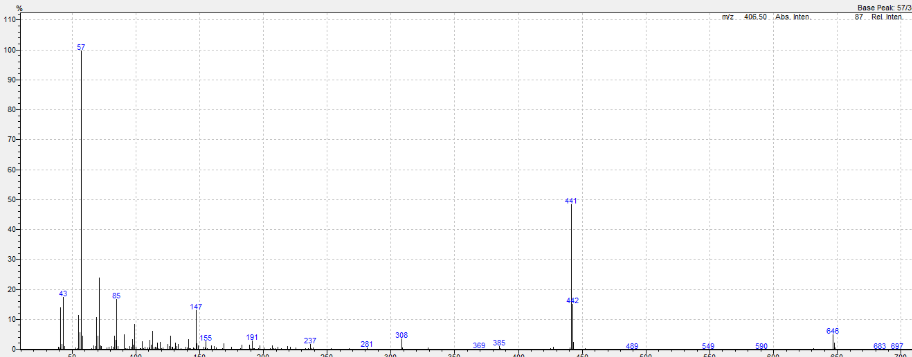
Figure 2. Typical mass fragmentation of an additive (e.g. Irgafos 168®).
Legal and toxicological assessment
Although NIAS are already mentioned in the Plastics Regulation, there are no legal limits for these substances in general. Manufacturers and distributors are obliged to carry out a (risk) assessment, even if the substances are not listed or previously known.
In practice, an initial assessment value of 10 µg/kg food (equivalent to 10 ppb) is initially used. This is based on regulations governing the migration of substances through a functional barrier, whereby unauthorised but intentionally added substances may be used behind a functional barrier provided that their migration is not detectable with a detection limit of 0.01 mg/kg food. This does not apply to substances that are classified as carcinogenic, mutagenic or toxic to reproduction (so-called CMR substances) and/or are present as nanoparticles. Since genotoxicity cannot be ruled out at first glance for initially unknown, unintentionally added substances, the estimated 10 µg/kg food is significantly higher than the value of 0.15 µg/kg food specified for this substance group in accordance with the TTC concept (Threshold of Toxicological Concern) and should therefore be regarded as a pragmatic measure rather than a concrete toxicological assessment value.
Substances that are detectable above this detection limit of 10 ppb (“10 ppb screening”) must then be identified and evaluated accordingly. In the case of listed substances, the assessment is often quite simple, as the respective specific migration limit (SML) can be used for the assessment. However, as NIAS are mostly unlisted substances, a risk assessment “in accordance with internationally recognised scientific principles” (Article 19 of the Plastics Regulation) is necessary. The respective substance is usually assessed on the basis of specific toxicological data and evaluations (e.g. by the EFSA, BfR), computer-aided models (in the absence of experimental data) or exposure estimates and genotoxicity tests.
Conclusion
NIAS are an unavoidable but controllable component of food contact materials. A thorough analysis strategy, combined with a scientifically sound toxicological assessment, is crucial to ensure compliance and product safety and to meet the requirements of EU legislation.
If you have any questions, please contact fcm@innoform.de. We will be happy to provide you with a quote for an NIAS analysis of your material.

